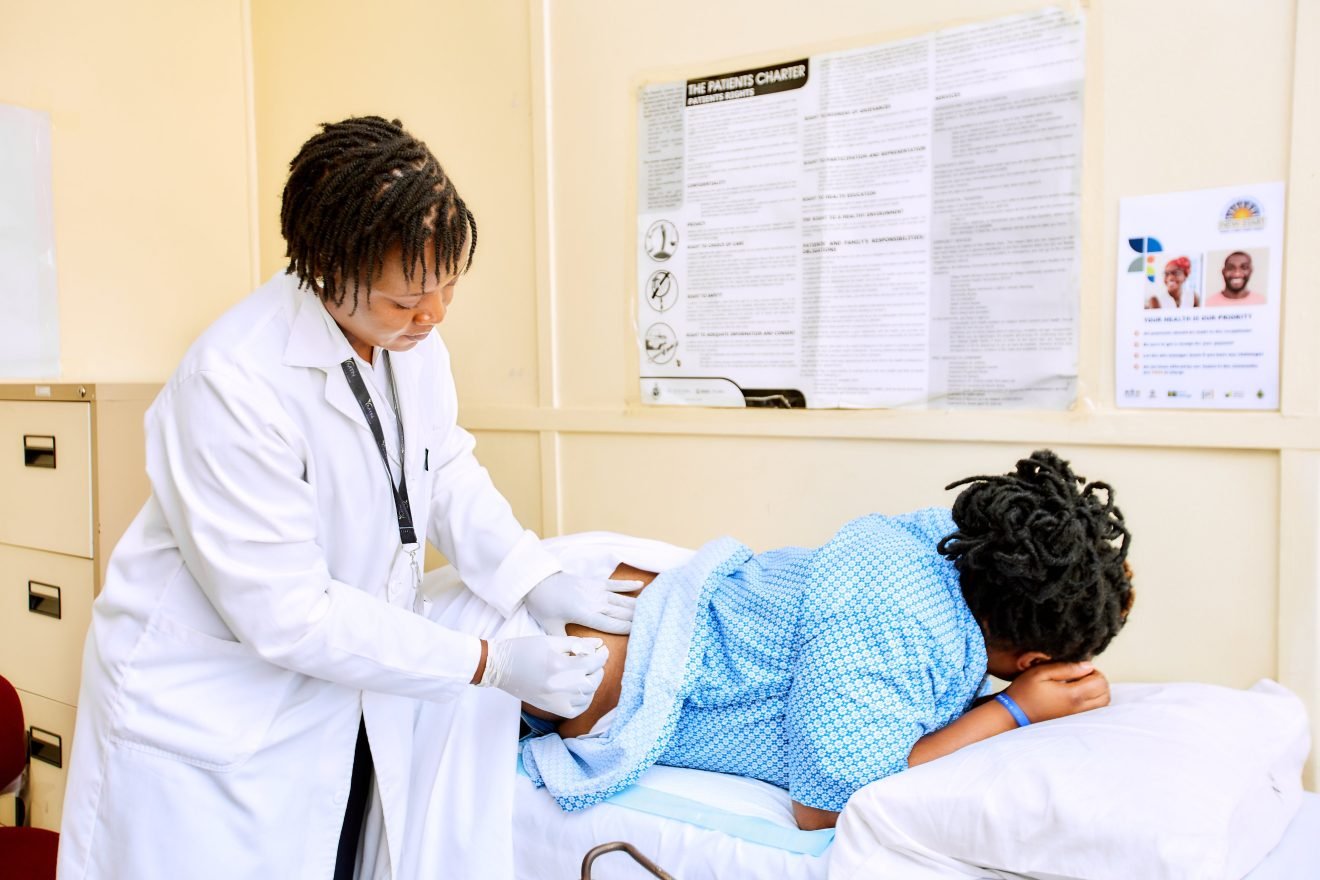
Medicines Control Authority of Zimbabwe (MCAZ) has approved Lenacapavir, a groundbreaking long-acting medicine used for HIV prevention, after completing an expedited review in just 23 days.In a statement released on November 22, MCAZ Director-General Richard Rukwata said the application for Lenacapavir, submitted by pharmaceutical company Gilead on 29 October 2025, went through a rigorous assessment before being cleared for use.He said the unusually swift approval reflects the medicine’s “major public health importance” in Zimbabwe and globally.“The Medicines Control Authority of Zimbabwe (MCAZ) announces the expedited approval of Lenacapavir, an innovative long-acting medicine for pre-exposure prophylaxis (PrEP) to prevent HIV-1 infection.“Lenacapavir was assessed and approved by the Registration Committee of the Authority under the expedited review process, as it is considered to be of major public health importance both in Zimbabwe and globally.“The application, submitted by Gilead on the 29th of October 2025, underwent a rigorous evaluation and was finalised in twenty-three (23) days, demonstrating the Authority’s commitment to ensuring timely access to essential, life-saving health products.”Lenacapavir is a long-acting PrEP option designed to prevent HIV-1 infection in adults and adolescents weighing at least 35kg.It is given as a subcutaneous injection every six months, following an initiation phase that includes one injection and two days of oral tablets.The treatment is intended for HIV-negative people at substantial risk of infection and must be used alongside safer sex practices, including condom use and behavioural risk-reduction measures.Health experts say the introduction of a long-acting injectable PrEP option could help improve adherence, particularly among communities that struggle with the daily pill regimen.“The rapid approval of Lenacapavir reflects MCAZ’s dedication to accelerating access to trusted, high-quality health products.“This milestone brings new hope for HIV prevention and reinforces our commitment to safeguarding public health.”The Authority used a regulatory reliance approach during the review process, drawing on scientific assessments from the World Health Organisation Prequalification programme to ensure a robust evaluation of the drug’s safety, efficacy and quality.TAGGED:ZimbabwePrevious ArticleControversial Operation Dudula Leader Hit With Teargas, Pepper Spray At G20 ProtestNext ArticleChinese National Arrested At RGMI Airport For Sexually Abusing Female Passenger On Emirates FlightLeave a ReplyCancel reply Medicines Control Authority of Zimbabwe (MCAZ) has approved Lenacapavir, a groundbreaking long-acting medicine used for HIV prevention, after completing an expedited review in just 23 days.In a statement released on November 22, MCAZ Director-General Richard Rukwata said the application for Lenacapavir, submitted by pharmaceutical company Gilead on 29 October 2025, went through a rigorous assessment before being cleared for use.He said the unusually swift approval reflects the medicine’s “major public health importance” in Zimbabwe and globally.“The Medicines Control Authority of Zimbabwe (MCAZ) announces the expedited approval of Lenacapavir, an innovative long-acting medicine for pre-exposure prophylaxis (PrEP) to prevent HIV-1 infection.“Lenacapavir was assessed and approved by the Registration Committee of the Authority under the expedited review process, as it is considered to be of major public health importance both in Zimbabwe and globally.“The application, submitted by Gilead on the 29th of October 2025, underwent a rigorous evaluation and was finalised in twenty-three (23) days, demonstrating the Authority’s commitment to ensuring timely access to essential, life-saving health products.”Lenacapavir is a long-acting PrEP option designed to prevent HIV-1 infection in adults and adolescents weighing at least 35kg.It is given as a subcutaneous injection every six months, following an initiation phase that includes one injection and two days of oral tablets.The treatment is intended for HIV-negative people at substantial risk of infection and must be used alongside safer sex practices, including condom use and behavioural risk-reduction measures.Health experts say the introduction of a long-acting injectable PrEP option could help improve adherence, particularly among communities that struggle with the daily pill regimen.“The rapid approval of Lenacapavir reflects MCAZ’s dedication to accelerating access to trusted, high-quality health products.“This milestone brings new hope for HIV prevention and reinforces our commitment to safeguarding public health.”The Authority used a regulatory reliance approach during the review process, drawing on scientific assessments from the World Health Organisation Prequalification programme to ensure a robust evaluation of the drug’s safety, efficacy and quality.TAGGED:Zimbabwe Medicines Control Authority of Zimbabwe (MCAZ) has approved Lenacapavir, a groundbreaking long-acting medicine used for HIV prevention, after completing an expedited review in just 23 days.In a statement released on November 22, MCAZ Director-General Richard Rukwata said the application for Lenacapavir, submitted by pharmaceutical company Gilead on 29 October 2025, went through a rigorous assessment before being cleared for use.He said the unusually swift approval reflects the medicine’s “major public health importance” in Zimbabwe and globally.“The Medicines Control Authority of Zimbabwe (MCAZ) announces the expedited approval of Lenacapavir, an innovative long-acting medicine for pre-exposure prophylaxis (PrEP) to prevent HIV-1 infection.“Lenacapavir was assessed and approved by the Registration Committee of the Authority under the expedited review process, as it is considered to be of major public health importance both in Zimbabwe and globally.“The application, submitted by Gilead on the 29th of October 2025, underwent a rigorous evaluation and was finalised in twenty-three (23) days, demonstrating the Authority’s commitment to ensuring timely access to essential, life-saving health products.”Lenacapavir is a long-acting PrEP option designed to prevent HIV-1 infection in adults and adolescents weighing at least 35kg.It is given as a subcutaneous injection every six months, following an initiation phase that includes one injection and two days of oral tablets.The treatment is intended for HIV-negative people at substantial risk of infection and must be used alongside safer sex practices, including condom use and behavioural risk-reduction measures.Health experts say the introduction of a long-acting injectable PrEP option could help improve adherence, particularly among communities that struggle with the daily pill regimen.“The rapid approval of Lenacapavir reflects MCAZ’s dedication to accelerating access to trusted, high-quality health products.“This milestone brings new hope for HIV prevention and reinforces our commitment to safeguarding public health.”The Authority used a regulatory reliance approach during the review process, drawing on scientific assessments from the World Health Organisation Prequalification programme to ensure a robust evaluation of the drug’s safety, efficacy and quality. Medicines Control Authority of Zimbabwe (MCAZ) has approved Lenacapavir, a groundbreaking long-acting medicine used for HIV prevention, after completing an expedited review in just 23 days. In a statement released on November 22, MCAZ Director-General Richard Rukwata said the application for Lenacapavir, submitted by pharmaceutical company Gilead on 29 October 2025, went through a rigorous assessment before being cleared for use.
He said the unusually swift approval reflects the medicine’s “major public health importance” in Zimbabwe and globally. “The Medicines Control Authority of Zimbabwe (MCAZ) announces the expedited approval of Lenacapavir, an innovative long-acting medicine for pre-exposure prophylaxis (PrEP) to prevent HIV-1 infection. “Lenacapavir was assessed and approved by the Registration Committee of the Authority under the expedited review process, as it is considered to be of major public health importance both in Zimbabwe and globally.
“The application, submitted by Gilead on the 29th of October 2025, underwent a rigorous evaluation and was finalised in twenty-three (23) days, demonstrating the Authority’s commitment to ensuring timely access to essential, life-saving health products.” Lenacapavir is a long-acting PrEP option designed to prevent HIV-1 infection in adults and adolescents weighing at least 35kg. It is given as a subcutaneous injection every six months, following an initiation phase that includes one injection and two days of oral tablets.
Read Full Article on Harare Times